MK-8591 –ISLATRAVIR: un nuovo NRTI a rilascio prolungato
Inviato: martedì 25 ottobre 2016, 0:45
Qui riporto l'articolo in lingua originale di un nuovo farmaco NRTI candidato ad essere somministrato in dosi a lunga azione.
Long-Acting Oral and Parenteral Dosing of MK-8591 for HIV Treatment or Prophylaxis
Author(s):
Jay Grobler1; Evan Friedman2; Stephanie E. Barrett1; Sandra L. Wood1; Wendy Ankrom1; Kerry L. Fillgrove1; Ming-Tain Lai1; Marian Gindy1; Marian Iwamoto3; Daria J. Hazuda1; for the MK-8591 Early Development Team
1Merck & Co, West Point, PA, USA;2Merck & Co, Rahway, NJ, USA;3Merck & Co, Kenilworth, NJ, USA
Abstract Body:
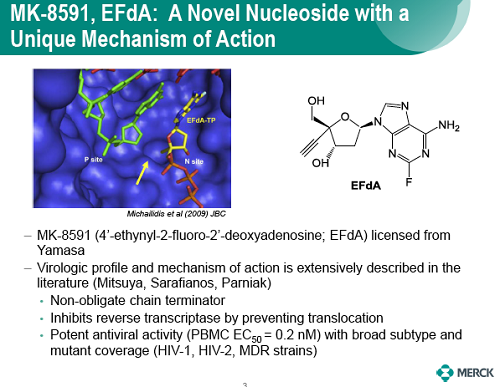
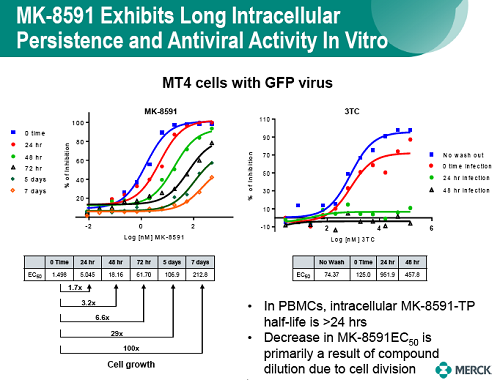
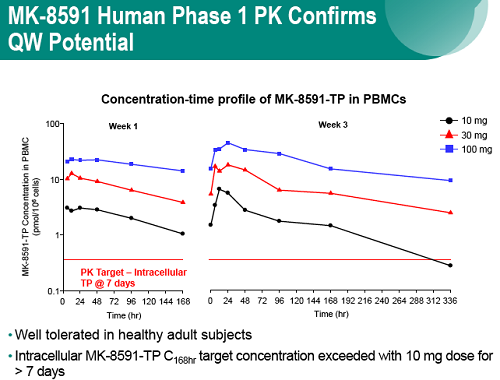
The potential to enhance adherence with less frequent dosing would represent a major advance for the treatment and prevention of HIV. MK-8591 is a nucleoside reverse transcriptase translocation inhibitor (NRTTI) with sub-nM potency that is in early stage clinical development. The phosphorylated anabolites of MK-8591, including the active triphosphate (MK-8591-TP), exhibit protracted intracellular persistence in human PBMCs and macrophages, protecting cells from infection in the absence of continued exposure in vitro. The potency, pharmacokinetic, and physical properties of MK-8591 are ideal for extended duration dosing.
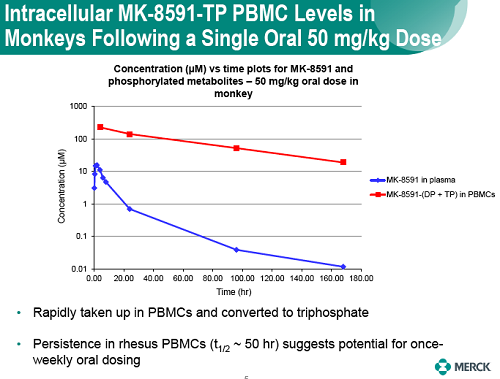
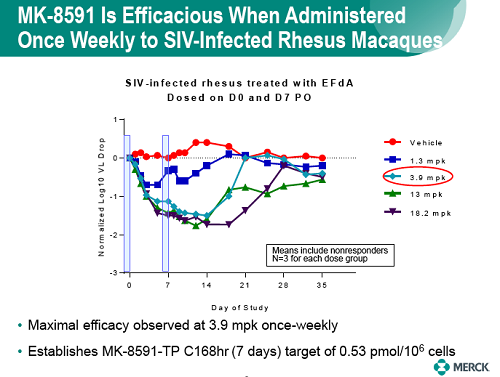
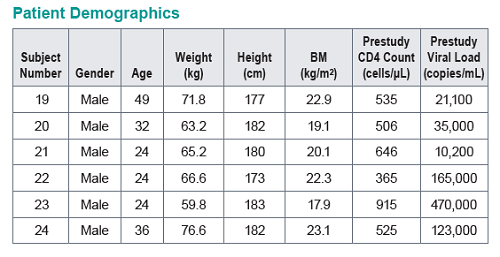
MK-8591 efficacy was evaluated in a SIVmac251-infected rhesus macaques dosed once weekly (QW) with 1.3 to 18.2 mg/kg. Doses were chosen on the basis of rhesus PK and intracellular NTP levels designed to match levels associated with antiviral efficacy in PBMCs. Plasma viral loads and MK-8591 concentrations (measured pre-dose through day 42) were used to a develop a PK/PD model and select ph1 doses for evaluation as QW oral dose in healthy subjects and enable dose selection for the development of long-acting parenteral formulations.
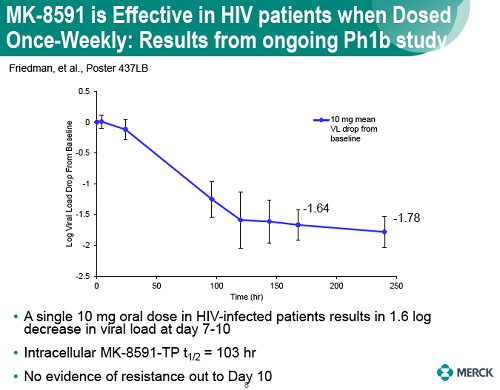
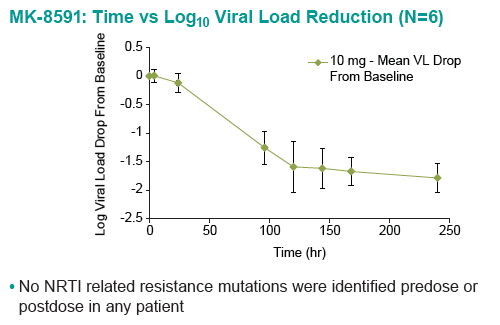
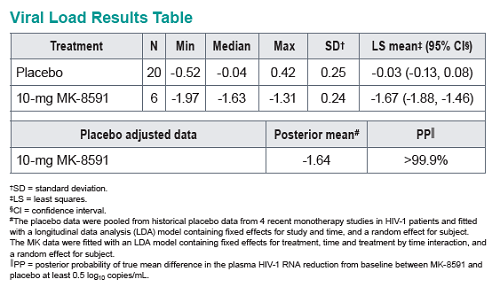
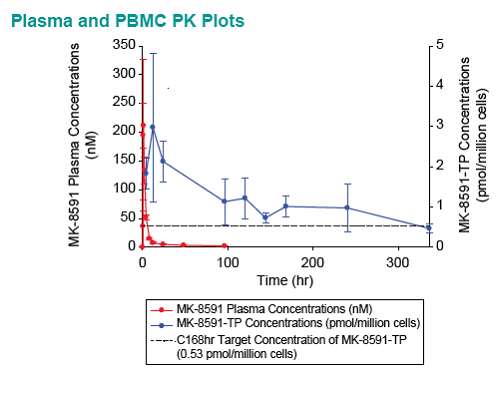
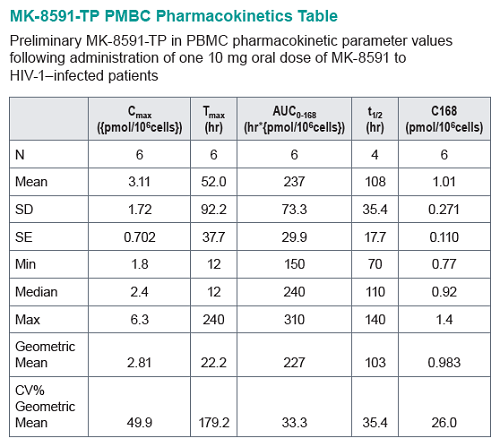
Baseline SIV viral loads in monkeys ranged from 106 to 108 copies per ml. After administration of QW doses of 3.9 to 18.2 mg/kg MK-8591, monkeys with viral loads 8 exhibited near maximal 2-log drops in viral loads and maintained suppression of viremia for at least 7days. MK-8591-TP concentrations of ≥0.53 pmol/106 PBMCs were associated with QW efficacy.
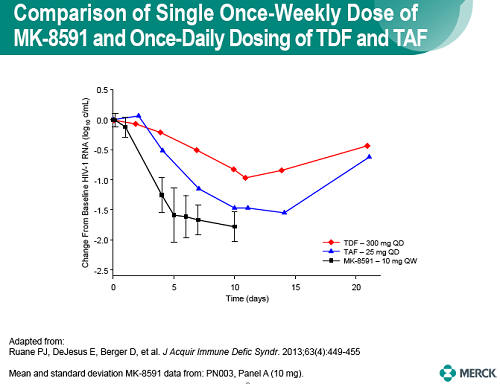
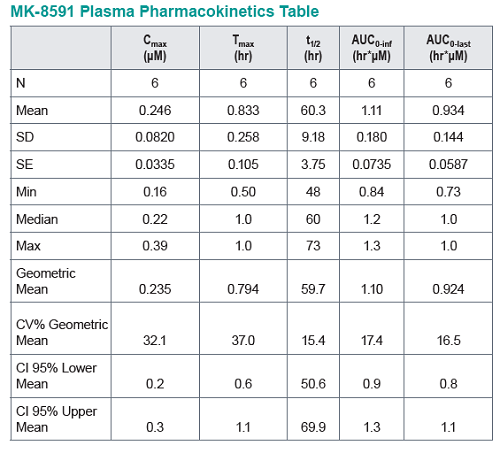
In healthy volunteers, single doses of 10 mg and greater were able to achieve these levels of MK-8591-TP for at least 7 days, suggesting the potential for QW antiviral efficacy at a low dose.
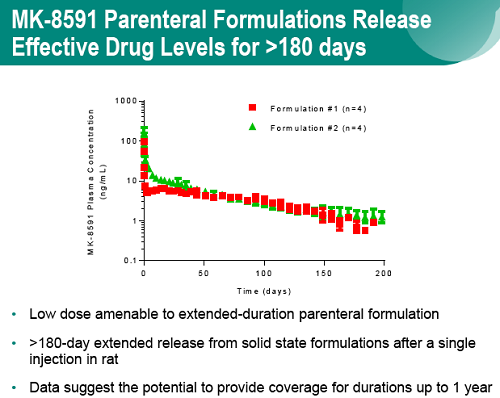
Finally, long-acting parenteral formulations of MK-8591 exhibited continuous, extended-duration drug release in rodents with MK-8591 plasma levels comparable to those achieved in rhesus and humans and duration of release exceeding 6 months.
The antiviral efficacy in an SIV rhesus macaque model when dosed QW together with the human PK data, suggest the potential for MK-8591 QW oral dosing in the clinic and a low dose compatible with delivery via long-acting formulations. MK-8591 QW oral and long-acting parenteral formulations with potential for six months or longer duration would represent a potential paradigm shift as a single agent for prevention of HIV infection or as a component of an extended dosing regimen for HIV treatment.
Session Number:
O-8
Session Title:
Drugs: From Discovery to Challenges in Clinical Use
Presenting Author:
Jay Grobler
Presenter Institution:
Merck & Co
Poster:
Poster to be submitted.
Se non erro si tratta della doravirina.
http://www.croiconference.org/sessions/ ... rophylaxis
Long-Acting Oral and Parenteral Dosing of MK-8591 for HIV Treatment or Prophylaxis
Author(s):
Jay Grobler1; Evan Friedman2; Stephanie E. Barrett1; Sandra L. Wood1; Wendy Ankrom1; Kerry L. Fillgrove1; Ming-Tain Lai1; Marian Gindy1; Marian Iwamoto3; Daria J. Hazuda1; for the MK-8591 Early Development Team
1Merck & Co, West Point, PA, USA;2Merck & Co, Rahway, NJ, USA;3Merck & Co, Kenilworth, NJ, USA
Abstract Body:
The potential to enhance adherence with less frequent dosing would represent a major advance for the treatment and prevention of HIV. MK-8591 is a nucleoside reverse transcriptase translocation inhibitor (NRTTI) with sub-nM potency that is in early stage clinical development. The phosphorylated anabolites of MK-8591, including the active triphosphate (MK-8591-TP), exhibit protracted intracellular persistence in human PBMCs and macrophages, protecting cells from infection in the absence of continued exposure in vitro. The potency, pharmacokinetic, and physical properties of MK-8591 are ideal for extended duration dosing.
MK-8591 efficacy was evaluated in a SIVmac251-infected rhesus macaques dosed once weekly (QW) with 1.3 to 18.2 mg/kg. Doses were chosen on the basis of rhesus PK and intracellular NTP levels designed to match levels associated with antiviral efficacy in PBMCs. Plasma viral loads and MK-8591 concentrations (measured pre-dose through day 42) were used to a develop a PK/PD model and select ph1 doses for evaluation as QW oral dose in healthy subjects and enable dose selection for the development of long-acting parenteral formulations.
Baseline SIV viral loads in monkeys ranged from 106 to 108 copies per ml. After administration of QW doses of 3.9 to 18.2 mg/kg MK-8591, monkeys with viral loads 8 exhibited near maximal 2-log drops in viral loads and maintained suppression of viremia for at least 7days. MK-8591-TP concentrations of ≥0.53 pmol/106 PBMCs were associated with QW efficacy.
In healthy volunteers, single doses of 10 mg and greater were able to achieve these levels of MK-8591-TP for at least 7 days, suggesting the potential for QW antiviral efficacy at a low dose.
Finally, long-acting parenteral formulations of MK-8591 exhibited continuous, extended-duration drug release in rodents with MK-8591 plasma levels comparable to those achieved in rhesus and humans and duration of release exceeding 6 months.
The antiviral efficacy in an SIV rhesus macaque model when dosed QW together with the human PK data, suggest the potential for MK-8591 QW oral dosing in the clinic and a low dose compatible with delivery via long-acting formulations. MK-8591 QW oral and long-acting parenteral formulations with potential for six months or longer duration would represent a potential paradigm shift as a single agent for prevention of HIV infection or as a component of an extended dosing regimen for HIV treatment.
Session Number:
O-8
Session Title:
Drugs: From Discovery to Challenges in Clinical Use
Presenting Author:
Jay Grobler
Presenter Institution:
Merck & Co
Poster:
Poster to be submitted.
Se non erro si tratta della doravirina.
http://www.croiconference.org/sessions/ ... rophylaxis